"Defining the baselines and standards for microplastics analyses in European waters (BASEMAN)" Work Packages:
Work Package 1
Reliable data on concentrations of MP and the composition of polymers present in the marine environment are lacking. This is because there are currently no standard operation protocols (SOP) for MP sampling and identification. Instead, a huge variety of different approaches from basic visual identification to highly sophisticated spectrometric techniques are applied during studies; precluding the general incomparison and evaluation of data. However, each of the techniques has its advantages and disadvantages and potentially provides additional information about the investigated particle (appearance, weathering, crystallinity). This work package aims to define the general limits (e.g., size, mass, polymer‐type) of the main methodologies currently used for the identification of MP by means of a MP test kit , which will be distributed among partners. The newest nano‐spectroscopic techniques will be evaluated for their ability to define an appropriate lower size limit of plastic particles (sub‐micron to nanoscale range). Furthermore, the changing (elemental) composition and conformation of pure polymers during ageing under natural/artificial marine conditions will be investigated through applied analytical techniques to ensure that these effects are appropriately incorporated into analytical approaches.
Objectives
- To develop and provide a MP reference kit for baseline definition and spiking of samples;
- To characterize the effects of artificial/natural weathering on reference MPs;
- To generate a spectra database (FT‐IR, Raman, Pyrolysis‐GCMS, HySpex) for pristine and weathered reference particles;
- To evaluate the strengths and limitations of different analytical techniques without interferences of environmental matrices;
- To develop methods with submicron capability in order to define a reasonable (methodological) lower size limit of MP.
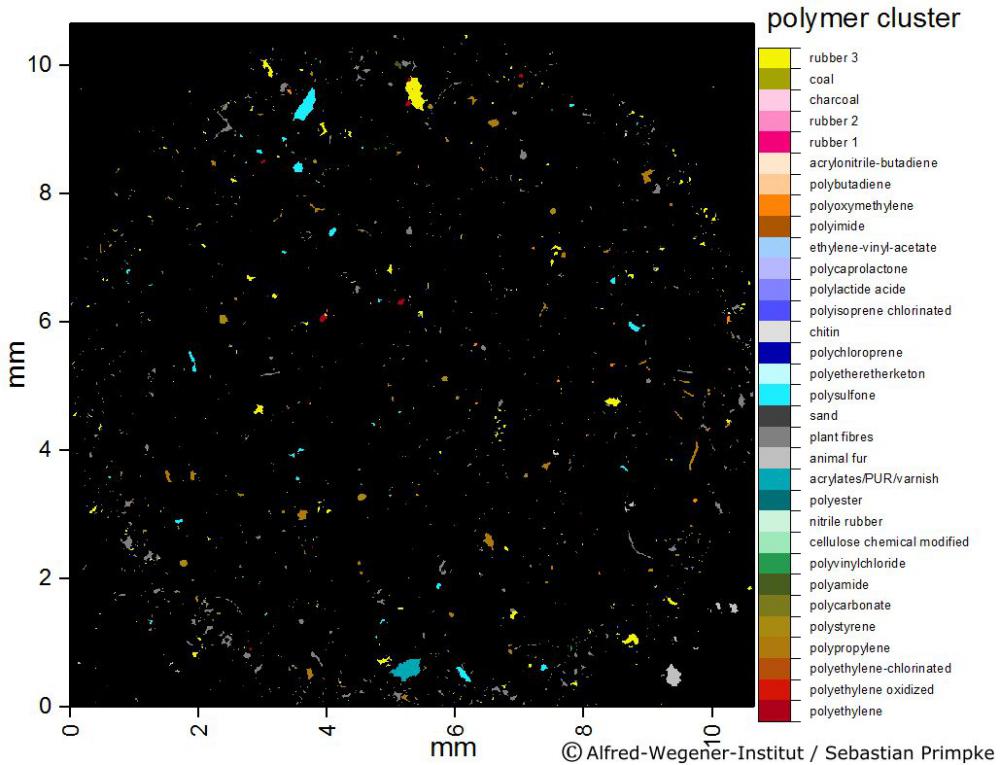
Work Package 2
Standardized samples will consist of three different samples in three replicates for each matrix (sediment, plankton, large biota). Samples will be spiked with the reference MP set (WP1 task 1.1). For this purpose natural plankton samples (with respect to plankton density and species composition), sediment‐samples (with respect to different grain size and organic load) and biota samples (fish, shellfish and lugworms) will be prepared and provided for all participating labs. All samples will be pre‐analyzed for their MP load before spiking and analytical characterization (details are given in the respective tasks). Standardized test samples without MP spiking will be provided for the inter‐method comparison approaches in WP3 tasks 3.2 and 3.3. Further standardized MP spiked test samples will be provided for labs participating in topic 2 of the call (detection) to be implemented in the analysis processes as blind samples.
Objectives
- To generate and provide standardized MP‐spiked test samples (sediment, plankton, biota) for inter‐lab comparisons;
- To generate and provide standardized test samples (sediment, plankton, biota) for inter‐method comparisons.

Work Package 3
A fundamental critical issue confronting current MP research is that the basis for an assessment of the environmental risk arising from MP is currently lacking. Reliable evaluation of MP concentrations and the composition of involved polymers in the marine environment are precluded by the lack of standard operation protocols (SOP) for MP detection. Thus, at the moment the comparability of data on MP concentrations is hampered by a huge variety of different methods, leading to the generation of data of extremely different quality and resolution. In WP3 we aim to address this problem by optimizing the purification and extraction approaches, and by comparing the workflows currently applied to MP analyses in sediments, plankton and biota. The latter approach is necessary since uneven and sub‐optimal analytical capacities (i.e.: access to FT‐IR or Raman; high standard chemical laboratories) exist in some laboratories responsible for analyzing MP pollution in the environment (for instance within the framework of the MSFD). Hence the synthesis of WP3 will allow these labs to critically evaluate their results with respect to the applied analytical workflow. All results will be critically evaluated within WP5.
Objectives
- To analyse MP‐spiked reference samples (sediment, plankton, biota) according to the current analytical workflows in the participating labs with respect to identification, quantification and sizing of MP;
- To analyse environmental samples (sediment, plankton, biota) provided by WP4 according to the current analytical workflows in the participating labs with respect to identification, quantification and sizing of MP;
- To optimize the extraction of MP from sediment;
- To optimize the purification of MP from sediment, plankton and biota in respect to matrix disintegration/removal and polymer preservation.

Work Package 4
The comparison and inter‐calibration of field and laboratory methods for measuring MPs is required for consistent analysis, and robust assessment and reporting of MP pollution levels in the marine environment. However, this is currently precluded due to the lack of standardized MPs sampling methodologies. The goal of WP4 is to develop robust methods for sampling seawater (both the water column and sea surface), sediments, and biota. This will produce Standard Operating Procedures (SOPs) for sampling sediments and seawater samples (from surface to the bottom) and biota (based on evaluations of field results in specific regions). In addition, the viability of alternative methods using platforms of opportunity will be evaluated in “pilot” regions. WP4 will provide WP3 with environmental samples. WPs 3 and 4 will be closely linked and the results will be critically evaluated within WP5. The planned exchange of researchers/students between these work packages is a key enabler of the integration of the entire project.
Objectives
- To develop standardized methods for sampling MPs in the water column and sediments;
- To identify marine biota species appropriate for MPs monitoring in European regions;
- To evaluate alternative methods for sampling MPs in the marine environment, particularly those deployable from platforms of opportunity;
- To intercalibrate sampling methods for MPs of varying size;
- To facilitate interlab comparisons and analyses of environmental samples of MPs;
- To develop validated conversion coefficients and harmonized reporting units for MPs.

Work Package 5
Internal administrative and financial management (including reporting and monitoring) will be coordinated by AWI. AWI will act as the intermediary between the funding agency administration and the administration of each participating organization. AWI will also co-ordinate the: Scientific management of the project; Compilation of progress reports; Monitoring of progress; and, External Progress reporting and evaluation. In the framework of BASEMAN huge amounts of data will be generated and maintained in a common web based format freely accessible to all participants. WP5 will provide and maintain this database by usage of Google‐docs, Google‐tables and Google‐drive. A critical synthesis of the outcome of the WPs dedicated to “Defining baselines for all relevant identification approaches” (WP1), “Inter‐lab and inter‐method comparisons” (WP3) and “Sampling methodologies for MPs in the marine environment: standardization, suitability and intercomparison” (WP4) will be pursued by specialized workshops that integrate work package participants with the rest of the consortium partners (workshops at start, mid‐term and end). The final aim will be to define with SOPs best practices for sampling (including de-vices, sample size, replicates) extraction, purification and identification of MP, but also recommendations for avoidance of contamination (best practice SOP). In addition to defining optimal procedures, it will also be necessary to define compromise approaches. This will facilitate those public authorities responsible for MP‐monitoring but without access to optimal facilities defined in best practice SOPs to undertake their responsibilities (best compromises SOP; e.g.: in the framework of MSFD). Hence, compromise approaches will provide guidelines, limits and reasonable thresholds for the commonly applied techniques for MP analyses (including basic microscopy). In this context, in the framework of the JPI‐O call “Ecological aspects of microplastics in the marine environment”, WP5 will inform all consortia addressing Topics 2 (Identification and quantification of microplastics) and 3 (Eco‐toxicological effects of microplastic – impact on marine organisms) with respect to MP identification and quantification. Further integration across the JPI‐O programme will be facilitated by several consortium partners who are also participating in Topic 2 or Topic 3 consortia. As specified in WP2, BASEMAN will provide reference blind samples for Topic 2 and 3 consortia upon request (to WP5) for consideration within relevant workflow analyses.
Objectives
- Ensure effective implementation and delivery of the BASEMAN project according to the description of work;
- To monitor, evaluate and report on progress and impacts;
- To integrate and critically review the results from WP1, WP3 and WP4;
- To organize and maintain databases for BASEMAN;
- To provide “best practice” and “best compromises” SOPs;
- To organize the kick off, interim and closing meetings for BASEMAN with participation of topic 2 and/or topic 3 consortia‐projects and stakeholders (upon invitation);
- To advise topic 2 and/or topic 3 consortia with respect to MP sampling, detection & analysis;
- To advice stakeholders with respect to MP sampling, detection & analysis;
- To organize and maintain outreach activities of BASEMAN.

